What happens if you mix lithium and aluminum together?
Answers
Answer:
hello
Explanation:
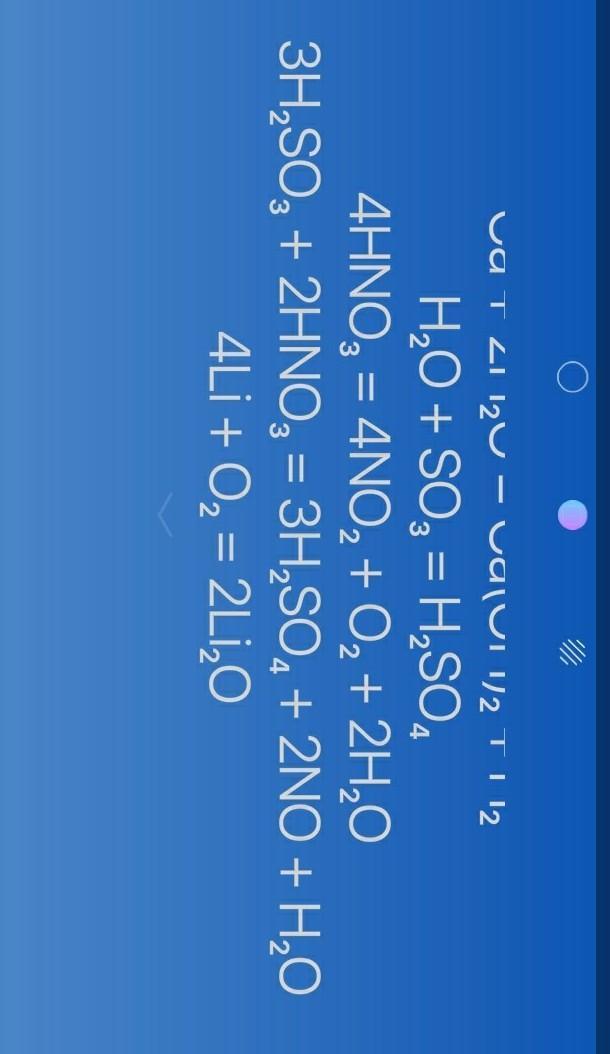
do you meant something like that

Related Questions
Brownian motion is
A. random movement of particles suspended in a fluid
B. movement of particles from an area of high concentration to low concentration
C. movement of particles from an area of low concentration to high concentration
D. random movement of smaller particles
Answers
Answer: A.
Explanation:
Brownian motion is the random motion of a particle as a result of collisions with surrounding gaseous molecules. Diffusiophoresis is the movement of a group of particles induced by a concentration gradient. This movement always flows from areas of high concentration to areas of low concentration.
Example: The movement of pollen grains on still water. Motion of dust motes in a room (though largely influenced by air currents).
What is the mass obtained of Ag from the reaction of 3 moles of AgCl
Given= Zn + 2AgCl —> ZnCl2 + 2Ag
Answers
Answer:
323.7g
Explanation:
Given parameters:
Number of moles of AgCl = 3 moles
Unknown:
Mass of Ag = ?
Solution:
We solve this problem by working from the known to the unknown;
The known here is AgCl;
From the balanced equation:
Zn + 2AgCl → ZnCl₂ + 2Ag
2 moles of AgCl will produce 2 moles of Ag
3 moles of AgCl will produce 3 moles of Ag
The mass of Ag will be;
Mass of Ag = number of moles x molar mass
Molar mass of Ag = 107.9g/mol
Mass of Ag = 3 x 107.9 = 323.7g
What would happen if there were 3 protons and 0 neutrons in the nucleus of an atom?
Answers
Answer:
the atom will be positife atom
1. Collect data: Create two water cycles using the Gizmo. Each cycle should have at least four steps and should begin and end at the same location. Choose any starting point from the list on the right. When the cycle is complete, choose the PATH tab and write the steps below.
Answers
Answer:
Cycle 1: Lake,atmosphere, clouds,rain,lake
Cycle 2: Atmosphere, clouds, rain, ocean
Convert 122 moles of Methane to liters?
Answers
susan calculates the heat energy released when water condenses using Q = mCΔT. explain why her calculation is incorrect?
Answers
You need to find the entropy of fusion in order to find this out.
What mass does 4.41 moles of cobalt have? Help!!!!
Answers
Answer:
260 g
Explanation:
Data obtained from the question include:
Mole of cobalt (Co) = 4.41 moles
Mass of cobalt (Co) =...?
The mole of a substance is related to its mass according to the following equation:
Mole = mass / Molar mass.
With the above formula, we can obtain the mass of 4.41 moles of cobalt as follow:
Mole of cobalt (Co) = 4.41 moles
Molar mass of cobalt = 59 g/mol
Mass of cobalt (Co) =...?
Mole = mass /Molar
4.41 = mass / 59
Cross multiply
Mass of cobalt = 4.41 × 59
Mass of cobalt = 260 g
Therefore, the mass of 4.41 moles of cobalt is 260 g
The most common physical state of the elements is
Answers
Answer: Solids, gases, or liquids
Explanation:
Matter can undergo physical and chemical changes called Phase Changes. Substances are classified based on physical states, often referred to as the States of Matter. At room temperature and pressure the common Physical States of elements are solids, gases or liquids.
Most are solids only 11 are gases and 2 are liquids
The most common physical state of elements in the periodic table is Solids as there are about 84 elements which exists as solids at room temperature and pressure in the periodic table
Elements in the periodic table can exist as one of the prominent physical states at room temperature and pressure.
These physical state may be;
SolidLiquidor GasElements which are solid at Room Temperature are about 84.At room temperature and pressure, only two elements are existent in liquid phase are: Bromine. Mercury.A look at the periodic table evidently tells that there are 11 elements in the table that exist in the gaseous state at room temperature and pressure.These elements are Hydrogen, Helium, Nitrogen, Oxygen, and the other Noble gases.
Read more:
https://brainly.com/question/21029213
what happens to the molecules as water freezes PLS HELP THIS IS DO TODAY
Answers
During freezing, water molecules lose energy and do not vibrate or move around as vigorously. This allows more stable hydrogen-bonds to form between water molecules, as there is less energy to break the bonds. ... Thus water expands as it freezes, and ice floats atop water.
When water molecule freezes, the water molecules have slowed down enough that their attractions fixed them into fixed positions.
What do you mean by freezing ?The term freezing is defined as a phase transition where a liquid converted into a solid when its temperature is lowered below its freezing point.
The freezing point is the temperature at which a liquid turns solid. In the freezer, for example, water-filled ice cube trays.
Water molecules freeze in a hexagonal pattern, with molecules further apart than when the water was liquid. It is worth noting that the molecules in ice would be vibrating.
Thus, When water freezes, the water molecules slow down to the point where their attractions arrange them into fixed positions.
To learn more about the freezing, follow the link;
https://brainly.com/question/26230908
#SPJ2
When exposed to very high temperatures, metals like iron can be turned into fluids that flow and can be poured into molds. What happens within the iron to allow it to flow?
A) The energy of the solid decreases, and the particles begin to collide with high frequency
B)The energy of the solid increases, and the particles begin to slide past each other
C)The solids collide at an increasing rate until reaching equilibrium energy
D)The particles vibrate about equilibrium in an effort to minimize energy
Answers
Answer: B) The energy of the solid increases, and the particles begin to slide past each other.
Explanation:
The energy of the solid matter is required to be increased so that molecules of the solid matter get separated from each other and an transition from solid matter to liquid matter occurs. The liquid components of the matter will slide past each other as they have obtain energy for fluidity. The liquid iron can be molded into any shape and the energy decreases considerably.
If carbon monoxide (CO) leaked into your home, would you predict that if would float to the
top of the room or stay near the floor? Answer the same question for carbon dioxide (CO2),
would it float or sink?
Answers
Answer: It will sink
Explanation:
To solve this we must be knowing each and every concept related to carbon monoxide. Therefore, carbon monoxide float to the top of the room.
What is carbon monoxide?When the carbon in fuels burns inefficiently, a toxic gas known as carbon monoxide is created. It is emitted both naturally—from forest fires or volcanic eruptions, for example—and artificially—through procedures. It is lighter than the air.
By attaching to and removing oxygen from myoglobin, carbon monoxide reduces the oxygen storage capacity in muscle cells, which is another way it upsets the body's processes. If carbon monoxide (CO) leaked into your home, carbon monoxide float to the top of the room. Numerous manufacturing and medicinal items employ carbon monoxide.
Therefore, carbon monoxide float to the top of the room.
To know more about carbon monoxide, here:
https://brainly.com/question/22530423
#SPJ5
In which situation is it better to have low friction?
Choose the correct answer.
a skydiver uses a parachute
a student walks down a hallway
a ladder leans against the wall
a hockey puck slides toward the goal
Answers
Answer: D- a hockey puck slides toward the goal
Explanation:
It needs to be able to slide easily and thats the correct answer on the test
An object accelerates 16.3 m/s2 when a force of 4.6 Newton’s is applied to it. What is the mass of the object?
A= .28 kg
B=74.98
C=3.8 kg
D= just to heavy to calculate
Answers
Answer:
A=0.28kg
Explanation:
FORCE=MASS * ACCELERATION
4.6=m*16.3
Then make m the subject.
m=4.6/16.3
m=0.28kg
What is the mass, in grams, of 0.450 moles of Sb?
Answers
Answer:
54.9 g
Explanation:
0.450 mol x 122g/mol
Relative and average atomic mass both describe properties of an element related to its different isotopes. Out of these two Relative atomic mas is more accurate. Therefore, 54.9 g is the mass in grams of 0.450 moles of Sb.
What is mass?Mass defines the quantity of a substance. It is measured in gram or kilogram. Average mass is the mass of atoms of an element that are isotopes. It can be calculated by multiplying mass of a isotope to natural abundance of that isotope.
Average atomic mass = (mass of first isotope× percent abundance of first isotope)+(mass of second isotope× percent abundance of second isotope)
Mass of Sb= number of moles of Sb ×Molar mass of Sb
=0.450 mol x 122g/mol
=54.9 g
Therefore, 54.9 g is the mass in grams of 0.450 moles of Sb.
To learn more about mass, here:
https://brainly.com/question/28704035
#SPJ6
What is the main difference between a chemical and a physical change? PLEASE HELP ASAP
Answers
Answer:
In a chemical change the nature of the substance changes and it is transformed, for example when a piece of wood is burned, the carbon is being transformed into carbon dioxide. A physical change is one where the nature of matter does not vary.
I hpoe help you.
Answer:
physical changes only change the appearance of a substance not it's chemical composition .chemical changes change the substance with a new chemical formula
Explanation:
Which is a way that the model of the atom became stronger through new experiments? (1 point)
a
New technology allowed scientists to show that the atom is indivisible.
b
It took new data to show that positive charge is not spread throughout an atom.
c
By deflecting alpha particles, Rutherford first observed the neutron.
d
The neutron could not be detected until Cathode–Ray Tubes were invented.
Answers
Answer:
Explanation:
Key points
J.J. Thomson's experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons.
Thomson's plum pudding model of the atom had negatively-charged electrons embedded within a positively-charged "soup."
Rutherford's gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus.
Based on these results, Rutherford proposed the nuclear model of the atom.
Identify whether each element is a halogen, a noble gas, or nonmetal only.
Astatine (At):
Nitrogen (N):
Krypton (Kr):
Chlorine (Cl):
Sulfur (S):
Answers
Answer:
Astatine: Halogen
Nitrogen: Non-Metal
Krypton: Non-Metal, Noble Gas
Chlorine: Non-Metal
Sulfur: Non-metal
Explanation:
if faucet drips 5 mL of water each minute, what is the volume of water dripped at the end of five minutes?
5 mL
25 mL
5 m³
25 m³
I apologize for being dumb XD
Answers
5 mL for the dripping rate times 5 for the minutes.
The answer would result being 25mL
Hope this helps! Stay safe!
A) saturated
B)desaturated
C)unsaturated
D)supersaturated
Answers
Answer:
No Its B Unsaturated becuase the line is slanted
Explanation:
How do you think the amount of a material affects its tendency to sink or float?
ASAP
Answers
Answer:
it dependes on the material
Explanation:
what is the material
Answer:
it doesnt
Explanation:
The amount of material does not reflect on its tendency to sink or float because that depends on density which is an intensive property.
What is the amount of mass in each given unit of volume?
Answers
Answer:
Each volumn is different and therefore has a specific and unique mass.
During the day, the earth is heated up by the sun. The land gets hotter than the sea because it takes less energy to increase the land's temperature than to increase the sea's temperature. The land heats the air near it. This air rises and is replaced by cool air from the ocean, creating a "sea" breeze that blows from the ocean to the land during the day. Which forms of heat transfer are described above as helping to create a sea breeze? A. convection only B. radiation only C. convection and conduction D. conduction and radiation
Answers
Answer:
C. convection and conduction
Explanation:
A sea breeze is a phenomenon that occurs during hot summer days when the sun transfers its heat to the earth surface by convection the land absorbs energy much easier than the seawater. As a result, the air above land will become warmer and as we know warmer air is lighter so this air will rise-up the air above the sea will now move from sea to the land to balance this air also there will be heat exchange between land air and the sea air by conduction and hence together they will create a phenomenon called a 'sea breeze'.
how many atoms in a chair
Answers
Answer:
100 trillion atoms are in one cell and so just find how many cells are in a chair and then take 100xthe number and you get your answer
Explanation:
There are uncountable atoms which are present in a chair depending on it's size.
What is an atom?An atom is defined as the smallest unit of matter which forms an element. Every form of matter whether solid,liquid , gas consists of atoms . Each atom has a nucleus which is composed of protons and neutrons and shells in which the electrons revolve.
The protons are positively charged and neutrons are neutral and hence the nucleus is positively charged. The electrons which revolve around the nucleus are negatively charged and hence the atom as a whole is neutral and stable due to presence of oppositely charged particles.
Atoms of the same element are similar as they have number of sub- atomic particles which on combination do not alter the chemical properties of the substances.
Learn more about atom,here:
https://brainly.com/question/30898688
#SPJ2
Pollinators, like bees, have a mutualistic relationship with plants. The pollinators carry pollen from plant to plant. What statement provides support for this as a mutualistic relationship?
A)The bees receive no benefit but are also not harmed. Only the plant receives benefits.
B)The bees provide protection for the flowers and the flowers protect the bees.
C)The bees deprive the flowers of valuable resources while pollinating the flowers.
D)The bees receive food from the nectar while the plants are pollinated.
Answers
Answer:
D)The bees receive food from the nectar while the plants are pollinated.
Explanation:
Bees and flowering plants have a mutualistic relationship where both species benefit. Flowers provide bees with nectar and pollen, which worker bees collect to feed their entire colonies. Bees provide flowers with the means to reproduce, by spreading pollen from flower to flower in a process called pollination.
Answer:
The bees receive food from the nectar while the plants are pollinated.
Explanation:
This is correct, but make sure it's for your answer choice, mine was A, but they switch them around.
list the elements symbols for the elements that are designated with neon green
Answers
H, C, N, O, P, S, Se
I saw this on a worksheet hope this helps!
Answer:
h,c,n,o,p those are the elements
How many neutrons make up one of these potassium atoms?
Answers
Answer:
20
Explanation:
Distinguish between organic acid and mineral acid
Answers
Answer:
Mineral acids are very important to chemical procedures. An organic acid is an organic compound with acidic properties. ... Generally, organic acids are weak acids and do not dissociate completely in water, whereas the strong mineral acids do.
Answer:
Organic Acids:
organic acids are weaker.organic acids always contain carbon, hydrogen and oxygen. organic acids are naturally occurring. organic acids are found in plants and animals.Examples: Methanoic acid, ethanoic acid, lactic acid etc.Mineral Acids:
Mineral acids are strong. Mineral acids are derived from one or more minerals found on Earth.Other than hydrogen, no other element is fixed in mineral acids.Example: Hydrochloric acid, sulphuric acidSe ha añadido un evaporador para una alimentación de 11500 kg/dia de zumo de pomelo de forma que evapore 3000 kg/dia de agua por un lado y se obtenga una disolución concentrado de 50% por el otro ¿ con qué concentración porcentual inicial se deberá alimentar el zumo? (m1 = m2 + m3) (%m1 = %m2 + %m3)
Answers
Answer:
37 %.
Explanation:
¡Hola!
En este caso, para el problema descrito, conocemos la corriente de entrada y la de salida del agua, por lo que podemos obtener el flujo de la corriente que contiene el zumo a la salida una vez el agua fue evaporada:
[tex]F_{sol}=11500kg/dia-3000kg/dia=8500 kg/dia[/tex]
Luego, por medio de un balance de zumo de limón en el evaporador en el cual la cantidad que entra es igual a la que sale con sus respectivas concentraciones:
[tex]x_z^{entra}*11500kg/dia=x_z^{sale}*8500kg/dia[/tex]
Como la concentración del zumo a la salida es del 50 % (0.50), la de entrada es:
[tex]x_z^{entra}=\frac{x_z^{sale}*8500kg/dia}{11500kg/dia} =\frac{0.50*8500kg/dia}{11500 kg/dia}\\ \\x_z^{entra}=0.37[/tex]
Que es igual al 37%.
¡Saludos!
GIVING 5 STARS AND BRAINLIEST IF YOU ANSWER FIRST AND CORRECTLY!! This is due today... so please help. Thank you.
QUESTION:
How many atoms does H2SO4 have?
Answers
Answer:
2 Hydrogen atoms, 1 Sulfur atom and 4 oxygen atoms.
Explanation:
H has a 2 next to it. S has no number assigned to it meaning it represents 1 atom. Lastly, O has a 4 next to it.
Hope this helps!!
Answer:
7
Explanation:
2 hydrogen atoms. 1 Sulphur atom. And 4 Oxygen atoms.
The superscript 2 and 4 show how many of the element are there.
Mark is in charge of making change at his family's garage sale on a hot summer day. He runs low on change and brings out a jar of pennies from the house. He keeps the lid off of the jar. Later in the day, he notices that the pennies are hot. Which statement best explains what happened?\
Answers
Answer:
This question is incomplete
Explanation:
The question is incomplete without the options. However, when a metal (in this case what the pennies are made of) is left in direct contact with the heat from the sun, it conducts this heat and becomes hot. This process is called thermal conduction. The thermal energy is readily accepted by the material in which the pennies are made of thereby increasing the thermal energy of the penny coin which then makes the pennies hot after being left under the sun.